 On day 7, she suddenly presented with chest pain, then lost consciousness, and no carotid pulse was found. In addition, creatine kinase (CK) and TnTsh were normal. After admission, we confirmed that her pacemaker was functioning normally (a pacing threshold of 0.6 V ms, a sensing threshold of 11.3–13.1 mV, and a lead resistance of 820 Ω). She received a secondary prevention of coronary heart disease. The concentration of hypersensitive cardiac troponin T (TnTsh, upper limit of normal = 14 pg/ml) was at a normal level (9.64 pg/ml). According to the ischemic symptom and previous history, the patient was diagnosed as having unstable angina. Electrocardiogram (ECG) on admission shows a sinus rhythm at 76 bpm, normal conduction, and low amplitude of T wave in multiple leads. Transthoracic echocardiography (TTE) showed a normal left ventricular size and function without regional wall motion abnormalities. On admission, her heart rate was 76 bpm (beats per minute) and her blood pressure was 131/67 mmHg. She was diagnosed as having SSS with 7.97 seconds of asystole and then received a dual-chamber pacemaker (Medtronic Inc., Minneapolis, MN, USA) implantation. One year ago, the patient was admitted to our hospital because of syncope. Then, coronary angiography revealed a severe stenosis of the left anterior descending (LAD), and a stent was implanted into the LAD. Six years ago, she was first admitted to our hospital and diagnosed with unstable angina. Her past history included coronary heart disease and sick sinus syndrome (SSS). Case reportĪ 71-year-old female presented to our hospital complaining of repeated chest pain. Herein, we report the rare case of failure pacing following an acute inferior wall myocardial infarction in a patient with an occlusion of the left circumflex (LCX) that supplied the right ventricular septal. 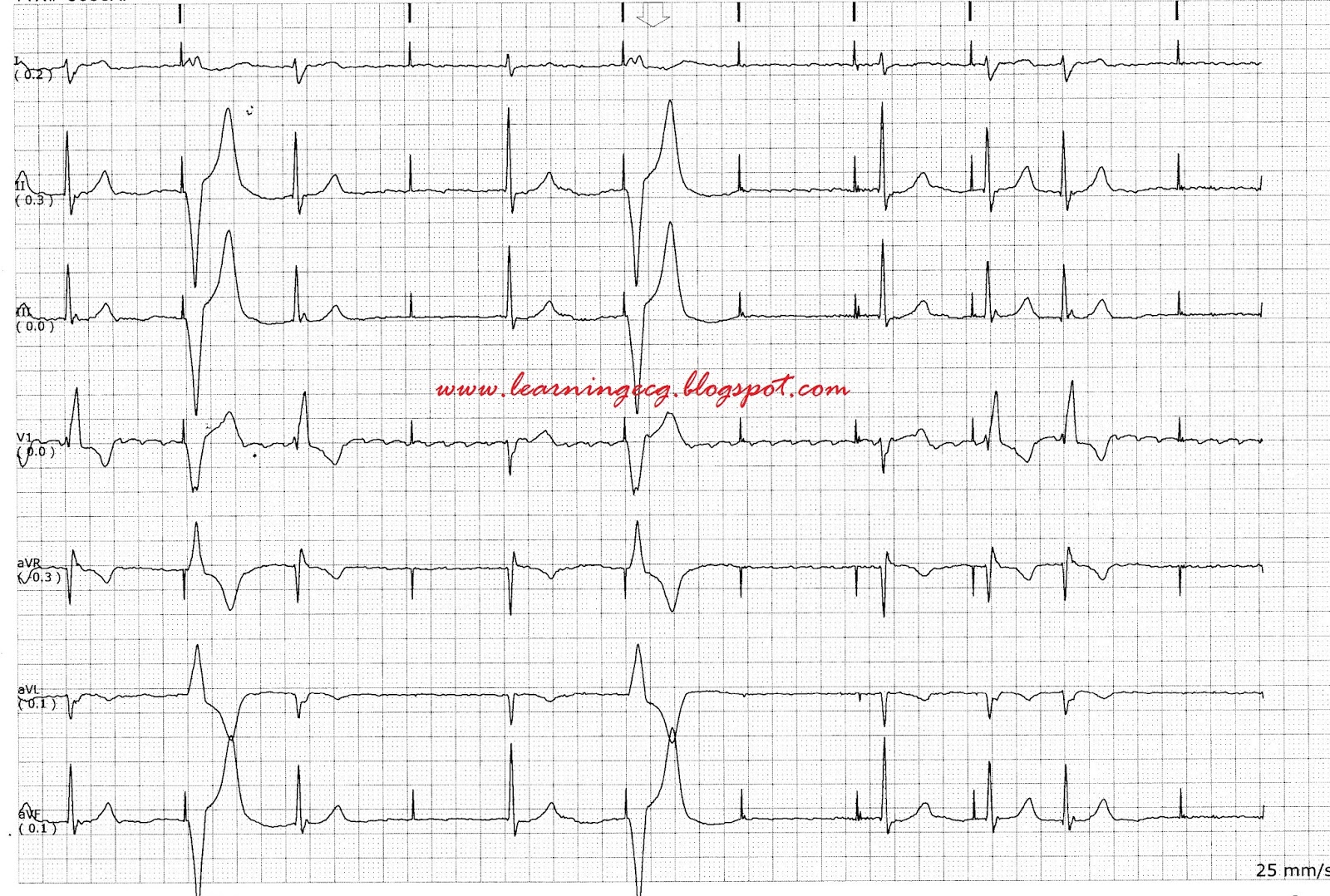
There are few reports describing the pacemaker lost atrial capture as a consequence of an AMI ( 2– 4). However, malfunction of a ventricular lead of a dual-chamber pacemaker following an acute myocardial infarction (AMI) is rare ( 1). There are several causes that contribute to the loss of a permanent pacemaker capture, such as battery depletion, lead dislodgement, and circuit problems. We conclude that the loss of the right ventricular lead pacing occurred in this case of acute coronary syndrome (ACS) induced by an LCX thrombus due to an LCX supplying the right ventricular septal. After reperfusion, the parameters of the right ventricular lead were gradually returned. 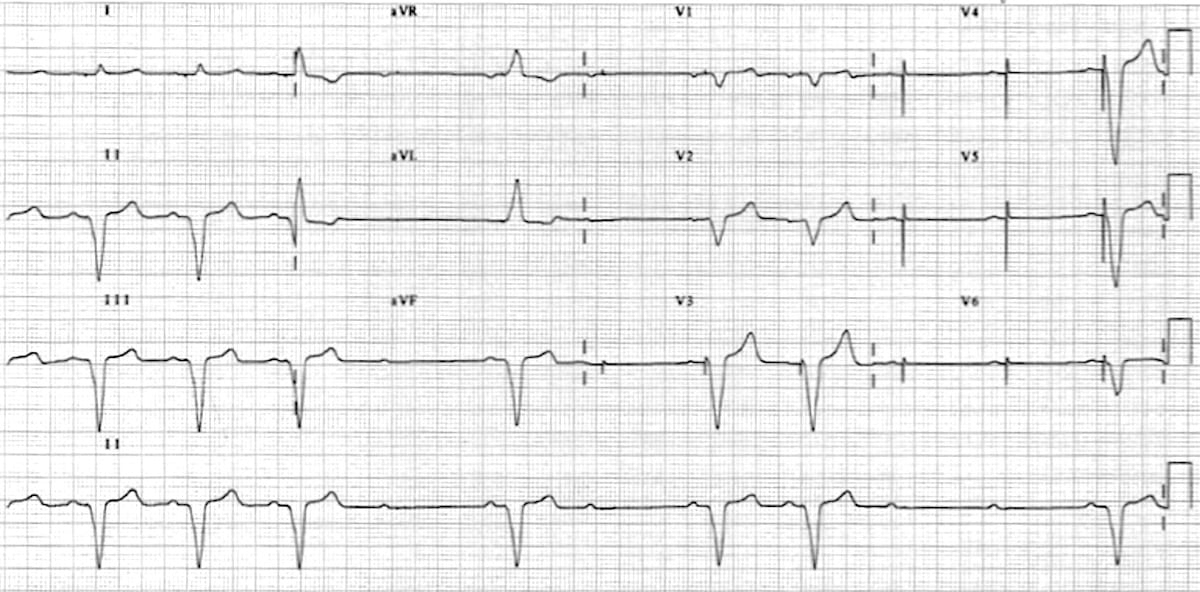 
Emergency coronary angiography revealed that a large filling defect remained due to an extensive thrombus in the proximal left circumflex (LCX) with thrombolysis in myocardial infarction (TIMI) grade 2 flow, and then a repeat thrombus aspiration was performed. Pacemaker lost capture was suspected and was later confirmed by a pacemaker check with a high pacing threshold and a low sensing parameter. On day 7, the patient suddenly suffered cardiopulmonary arrest due to an inferior ST segment elevation myocardial infarction (STEMI). 
Department of Cardiology, The Second Xiangya Hospital of Central South University, Changsha, ChinaĪ 71-year-old female with a dual-chamber pacemaker presented to our hospital complaining of repeated chest pain.Increasing the energy output of programmable pacemakers, in specific circumstances, or insertion of a temporary pacemaker capable of delivering higher energy output than the implanted generator may also be successful.Zhihong Wu † Jianjun Tang † Qingyi Zhu Lin Hu Zhenjiang Liu Xuping Li Qiming Liu Shenghua Zhou Mingxian Chen * Acute increases in pacing threshold immediately following pacing system implantation can be treated with corticosteroids. Management of failure to capture is directed at removing the inciting cause and attempting to lower the pacing threshold with isoproterenol, if the situation is urgent. Patients with pacemakers who require antiarrhythmic agents or who have coincident metabolic or electrolyte disturbances should be observed closely for failure to capture. Commonly used antiarrhythmic drugs (quinidine and procainamide) also increase pacing threshold. Electrolyte and metabolic abnormalities, especially hyperkalemia, alkalosis, acidosis, and hyperglycemia, increase the pacing threshold. Physiologic variations due to changes in autonomic tone (including eating, sleeping, and exercise) account for day-to-day fluctuations in pacing threshold. The acute local tissue reaction at the electrode-myocardium interface accounts for early threshold rises and may be minimized by choice of specific electrodes (steroid-eluting or carbon-tip). Many factors affect the threshold for myocardial stimulation in patients with cardiac pacemakers. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
